For medical device manufacturers, delivering more value for patients and clinicians is the engine of business growth. Value can follow incremental improvements to a device, or value can emerge by using analytics on data aggregated from the device and related sources. This emergent value can increase your return on investment in new and older products and boost the efficiency of the entire product lifecycle – from inception to postmarket surveillance.
Despite this opportunity, MedTech organizations often face challenges in turning a vision for data- and analytics-enhanced products into reality. The challenges include:
- Reliable data integration for medical devices that connect to various systems and data sources
- High costs in time and money to clean data from diverse sources for use in analytics and other applications
- Integrating insights from the data back into the familiar workflows of clinicians so they and their patients can benefit from them
Medical device manufacturers are finding solutions to these challenges with InterSystems technology and partnership. Our advanced cloud-first data platform, InterSystems IRIS for Health™, empowers you to harness interoperability standards, multi-model data management, natural language processing, analytics, and machine learning to cost-effectively drive innovation and value.
The following uses cases illustrate a few ways you can work with InterSystems for data-driven innovation and the creation of value-added services for your organization.
Use Case: Postmarket Study of Cardiac Device Performance
A major medical device manufacturer already had success in the market with one of its IoT enabled implantable monitoring devices. Telemetry from the device helped them monitor its performance for postmarket surveillance. The problem was that the data collected did not tell the manufacturer or clinicians how the device data related to patient outcomes, or which patients responded best to the device.
The manufacturer saw an opportunity to address these issues by combining and correlating three diverse data sources: time series from multiple versions of an implanted device, qualitative input from an electronic Patient Reported Outcome (ePRO) questionnaire on the patient’s smart phone, and clinical data from the patient’s electronic health record (EHR). But first, data from these sources, in multiple formats, had to be rationalized into one consistent representation for analysis.
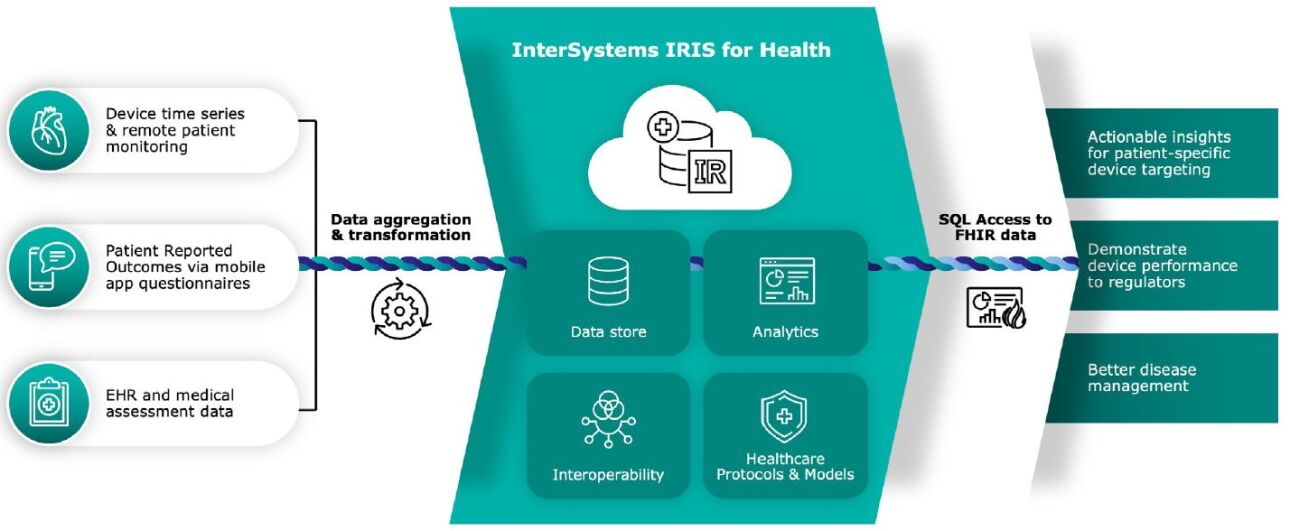
InterSystems Combines FHIR and SQL to Simplify Data Access for Analytics
The medical device manufacturer partnered with InterSystems to use InterSystems IRIS for Health data platform to aggregate and, when necessary, transform all the data into one consistent HL7® FHIR® (FHIR) representation. Solution developers extended a FHIR data model using InterSystems IRIS for Health to incorporate device data, performance data, PROMS, and time series, and prepare it for access with InterSystems IRIS for Health FHIR SQL Builder. With the FHIR SQL Builder, data scientists can use the SQL that they already know to access FHIR data. This sophisticated projection tool creates custom SQL schemas using data in an InterSystems IRIS for Health FHIR repository without moving the data to a separate analytics repository.
Because the FHIR data remains in an InterSystems IRIS for Health database, as opposed to a separate product for analytics, it is always up to date and ready for use. This saves medical device manufacturers the high cost, in time and money, of copying large amounts of data to a separate analytics repository and maintaining it. Analyzing all the data, this manufacturer is discovering:
- Insights to help target the device to the types of patients who respond best to its use
- Ways that its customers can improve disease management
- Statistics for regulators that demonstrate device effectiveness and reduced risk
Use Case: Actionable Insights from Interoperable Device Data and Analytics
How can a medical device manufacturer best leverage the volume of data generated by their devices to help their customers understand operational patterns across the care continuum? One such manufacturer is creating a “digital services” offering that enables their healthcare customers to independently run queries and analyze the data collected by medication dosage devices. The goal is to understand and improve drug dosage, usage, and inventory patterns.
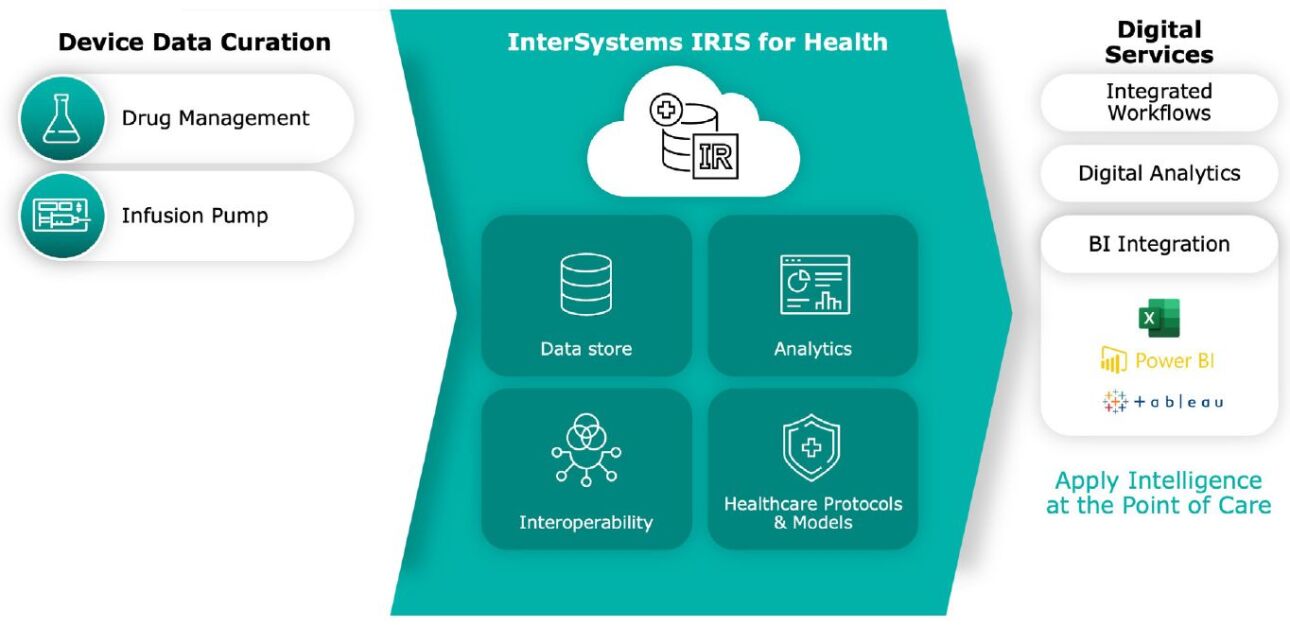
The solution uses the data transformation and integration power of InterSystems IRIS for Health data platform and InterSystems IRIS Adaptive Analytics. Adaptive Analytics creates a common semantic layer, for use by healthcare analysts and their preferred analytics tools, above the large and diverse data sets collected for each healthcare organization.
The device manufacturer is gaining a new source of revenue and differentiation from competitors by leveraging data its devices already produce. Its customers are providing a curated data set and common semantic layer across devices for exploration by their analysts, using their preferred business intelligence (BI) tools such as Tableau or PowerBI. They can look at the data from multiple dimensions and combine it with other data to answer questions such as:
- What is the pattern of administered drug dosages over time and by location?
- How much of a drug’s dosages have been wasted versus completely used?
- Do we have enough critical medications on hand?
- Are alarms effective, or are they being overridden?
- Are we meeting our critical key performance indicators and safety standards?
Use Case: Connecting Remote Point of Care Diagnostics to Health Records
A manufacturer of point of care (POC) diagnostic devices found an unmet customer need specific to clinician offices or mobile care locations in underserved communities. These remote care settings often lack internet access and the POC diagnostic devices, despite supporting connectivity and data integration standards, were not always connected directly to an EHR or to the device manufacturer’s cloud analytics platform. So, the broader care team providers were often unaware of the diagnostic results. Third-party software is available to address these issues, but it comes at a significant and unsustainable cost for these practices as the number and variety of devices grows.
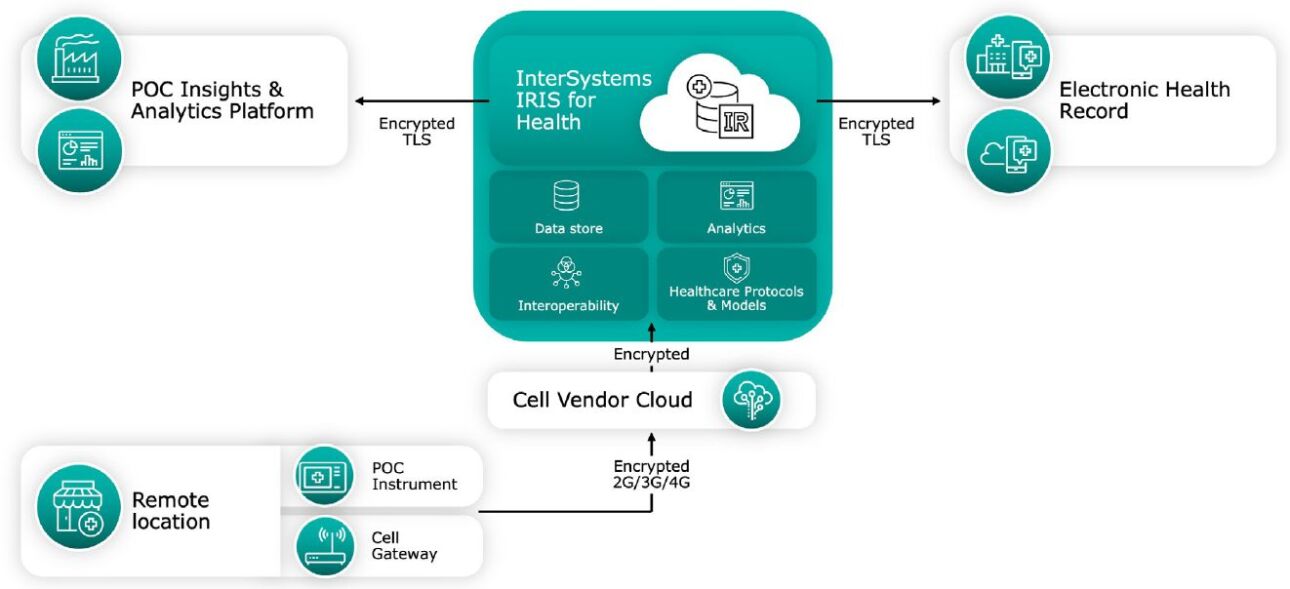
Simplified and Flexible Interoperability for Remote Testing Locations
Using a cellular data network gateway and InterSystems IRIS for Health data platform, the medical device manufacturer can develop a cost-effective solution that eliminates the need for third-party software and enhances the value of its POC diagnostics.
The POC devices will feed data to the cellular gateway, with InterSystems IRIS for Health providing the necessary data transformation and integration capabilities. InterSystems IRIS for Health will connect multiple remote devices to the manufacturer’s POC cloud analytics platform, for visibility into device performance, and send patient testing results to an EHR. This architecture is another example of product differentiation using InterSystems IRIS for Health, as the device manufacturer creates a ready-to-use option for clinician offices to connect to any EHR or laboratory information system.
Learn More
These use cases all benefit from having a single platform and database that product development teams can use to create complete solutions. InterSystems IRIS for Health leverages data from multiple, diverse sources to address the simplest to the most complex data and application challenges in MedTech. You can learn more about our support for MedTech innovation at InterSystems.com/MedTech.




























